The Lundquist Institute (TLI) start-up company, Vitalex Biosciences, has been awarded an SBIR Phase 2 grant from the National Institute of Allergy and Infectious Diseases (NIAID) at the National Institutes of Health. The grant is for Vitalex’s VX-01, a monoclonal antibody (mAB) program targeting the debilitating indication of the fungal disease, mucormycosis. This serious fungal infection often occurs in people who are immunocompromised and is spreading throughout the world. Mucormycosis is only curable when diagnosed in its early stages.
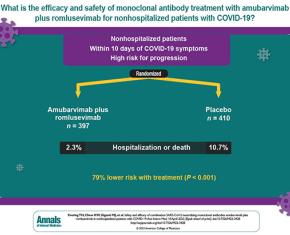
The study showed that antibody combination provides strong protection against severe COVID-19 in large international trial. A treatment combining two antibodies against the coronavirus SARS-CoV-2 strongly protected high-risk people with early COVID-19 symptoms from hospitalization and death in an international Phase 2/3 clinical trial. The trial enrolled more than 800 non-hospitalized patients with COVID-19 at high-risk of progression of the disease in the United States and five other countries.